计算题:欲配制c(1∕2EDTA)=0.10mol∕L乙二胺四乙酸二钠(EDTA二钠)溶液1000mL,需用EDTA二钠多少克?
正确答案:
1∕2EDTA二钠的摩尔质量为186.12g∕mol,设需EDTA二钠的质量为Wg,则:
W=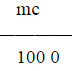 •V=
•V=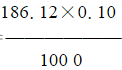 ×1000=18.612g
×1000=18.612g
配制c(1∕2EDTA)=0.10mol∕L乙二胺四乙酸二钠(EDTA二钠)溶液1000mL,需用EDTA二钠18.612克。
W=
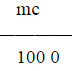 •V=
•V=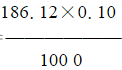 ×1000=18.612g
×1000=18.612g配制c(1∕2EDTA)=0.10mol∕L乙二胺四乙酸二钠(EDTA二钠)溶液1000mL,需用EDTA二钠18.612克。
答案解析:有

微信扫一扫手机做题
