计算298K下,CO(g)和CH3OH(g)的标准生成焓,并计算反应CO(g)+2H2(g)=CH3OH(g)的标准焓变。已知如下燃烧焓数据: (1)ΔcHm$(298K)(CH3OH(g))=-763.9kJ·mol-1 (2)ΔcHm$(298K)(C(s))=-393.5kJ·mol-1 (3)ΔcHm$(298K)(H2(g))=-285.8kJ·mol-1 (4)ΔcHm$(298K)(CO(g))=-283.0kJ·mol-1
正确答案:
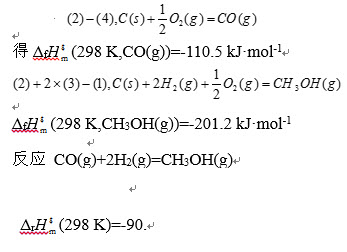
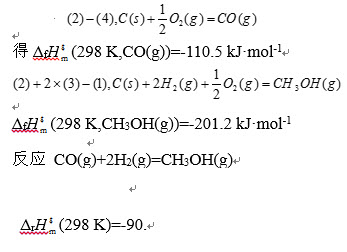
答案解析:有

微信扫一扫手机做题
